The Clock on Your Patent
A plain-English guide to US patent term, why it almost never runs the full 20 years, and how the rules compare around the world.


Imagine you spend a decade perfecting a drug that treats a rare blood cancer. You file your patent application the day you first describe the invention. Five years later — after mountains of paperwork, examiner interviews, and response deadlines — the patent finally issues. You now have a legal monopoly on your invention. But for how long? The answer is almost certainly not what you expected, and the gap between expectation and reality has enormous commercial consequences.
Patents are, at their heart, a time-limited bargain: society grants an inventor exclusive rights in exchange for full public disclosure of the invention. Once the clock runs out, the invention enters the public domain and anyone can freely use, make, or sell it. Understanding exactly how long that clock runs — and what can speed it up or slow it down — is therefore one of the most practically important questions in intellectual property.
The 20-Year Baseline
For any US patent application filed on or after June 8, 1995, the statutory rule is straightforward on its face. The USPTO's own Manual of Patent Examining Procedure (MPEP) sets it out in unambiguous terms:
"A patent granted on an application filed on or after June 8, 1995 shall have a term that ends 20 years from the date on which the application for the patent was filed in the United States, or, if the application contains a specific reference to an earlier filed application or applications under 35 U.S.C. 120, 121, or 365(c), from the date on which the earliest such application was filed."
MPEP § 2701 — Patent Term
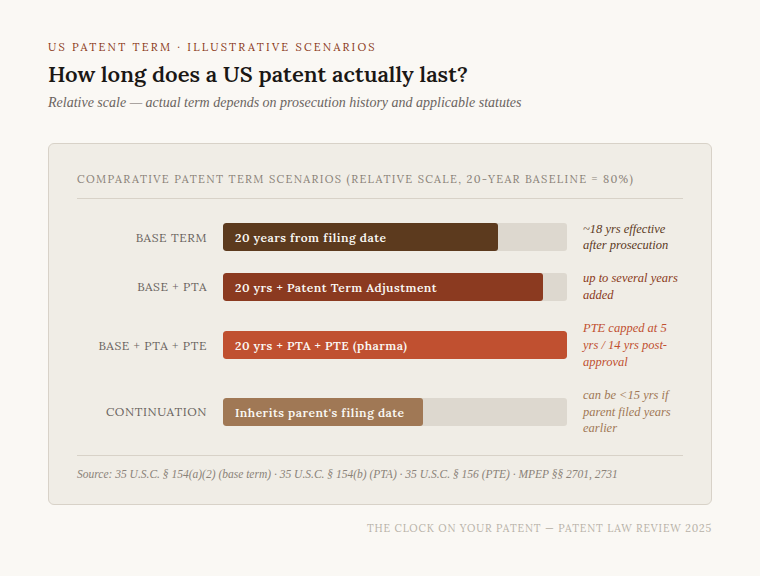
That "20 years from the earliest filing date" phrasing carries a sting for inventors who use continuation or divisional applications. In patent prosecution, it is common practice to file a continuation to claim variations not captured in the parent application, or to strategically broaden or narrow claim scope. Each of those continuation applications inherits the priority date of the original parent — and so the 20-year countdown began on the day you filed that parent, not the day you filed the continuation. A patent issued on a continuation application filed fifteen years after the parent has only five years of remaining term on the date it issues.
Layperson's analogy: Think of the 20-year clock as a parking meter you start the moment you pull into the spot. It doesn't reset each time you feed the meter — filing a continuation is like paying more quarters, but the original expiry time never moves.
Importantly, the patent term runs from the filing date, not from the issue date. Before 1995, US patents ran for 17 years from the date of issuance. The change aligned the US with international practice under the TRIPS Agreement and the Patent Law Treaty, but it created a new problem: delays during prosecution now directly eat into the term you actually get to use.
Patent Term Adjustment (PTA)
Congress recognised that entitling the USPTO to take as long as it wished to examine an application would be fundamentally unfair — the agency's slowness would be the inventor's loss. The American Inventors Protection Act of 1999 therefore created the Patent Term Adjustment (PTA) mechanism, which adds days to the patent's term to compensate for certain USPTO-caused delays. The statutory framework is codified in 35 U.S.C. § 154(b), and the MPEP explains the three distinct categories of qualifying delay:
"35 U.S.C. 154(b) provides for an adjustment of the term of a patent if the issuance of the patent was delayed due to (A) delays due to the failure of the Office to take certain actions within prescribed time frames (the 'A delay'), (B) delays due to the failure of the Office to issue a patent within 3 years of the actual filing date of the application in the United States (the 'B delay'), or (C) delays due to interference or derivation proceedings, secrecy orders, or appeals to the Patent Trial and Appeal Board or Federal Circuit (the 'C delay')."
MPEP § 2731 — Basis for Patent Term Adjustment
Type A Delays — The Office's failure to act promptly
Type A delays are generated when the USPTO misses specific internal deadlines: failing to mail a first Office Action within 14 months of filing, failing to respond to a reply from the applicant within four months, or failing to issue a patent within four months of the issue fee payment, among others. Each day the USPTO exceeds these windows is a day added back to the patent term.
Type B Delays — The three-year pendency guarantee
Congress went further and promised that every applicant is entitled to a patent issuing within three years of the US filing date — barring applicant-caused delays. If the USPTO fails to issue the patent within that window, every extra day of pendency becomes a PTA day. This is particularly important for complex biotechnology and pharmaceutical applications, where prosecution commonly stretches four, five, or even seven years.
Type C Delays — Interference and appeals
If prosecution is stalled because of an inter partes proceeding, a secrecy order imposed for national security reasons, or a successful appeal to the Patent Trial and Appeal Board (PTAB) or the Federal Circuit, those delay days also count in the applicant's favor.
The applicant-delay deduction
The PTA calculation is not entirely one-sided. The MPEP is clear that any PTA accrued by the USPTO is reduced by delays attributable to the applicant:
"The period of adjustment ... shall be reduced by a period equal to the period of time during which the applicant failed to engage in reasonable efforts to conclude prosecution of the application."
MPEP § 2733 — Reduction of Patent Term Adjustment
Practically, this means that every day an applicant takes beyond three months to respond to an Office Action reduces the PTA award by one day. Filing an RCE (Request for Continued Examination) — a very common prosecution tool — also resets the Type B clock, often wiping out accumulated B delays entirely.
"A PTA of several hundred days is routine in pharmaceutical prosecution. In extreme cases, applications pending a decade or more can emerge with PTAs exceeding 1,500 days."
Patent Term Extension (PTE) — The Pharmaceutical Safety Net
PTA handles delays caused by the patent office. But for pharmaceutical and medical-device companies, there is a second, entirely separate source of lost commercial time: the regulatory approval process at the FDA. A drug patent might issue on day one of clinical trials, but the drug cannot legally be sold until the FDA approves it — a process that commonly takes five to ten additional years. The Hatch-Waxman Act of 1984 created the Patent Term Extension (PTE) mechanism to address this.
Under 35 U.S.C. § 156, a patent covering a product subject to regulatory review — primarily drugs, biological products, medical devices, food additives, and animal drugs — may be extended for up to five years, subject to a cap: the total remaining term after PTE cannot exceed 14 years from the date of product approval. The extension covers only one patent per regulatory approval, and the patent must be the first to claim the approved product or method of use.
PTE and PTA are distinct mechanisms and are not straightforwardly additive. A patent eligible for both undergoes a careful accounting to ensure the combined result does not exceed the statutory maximums. For blockbuster drugs, the commercial value of even one additional year of exclusivity can run into the billions of dollars, making PTE applications intensely scrutinised and frequently litigated.
What Else Affects Your Patent Term?
Terminal disclaimers
When an applicant owns multiple related patents that could be seen as claiming the same invention (a problem called "obviousness-type double patenting"), the USPTO requires the applicant to file a terminal disclaimer — a formal surrender agreeing that the later patent will expire no later than the earlier one. Terminal disclaimers also create a "common ownership" requirement: the two patents must remain under common ownership throughout their enforced lives. The practical effect can be severe; a continuation patent filed years after the parent may be forced to expire on the same day as the parent, negating much of the strategic value of the continuation.
Maintenance fees
Even within the patent term, a patent does not remain in force automatically. US patents require maintenance fees paid at 3.5, 7.5, and 11.5 years after issuance. Failure to pay any maintenance fee causes the patent to lapse — it effectively dies early. Small entities and micro-entities pay reduced fees, but the obligation is absolute.
Prior art and validity challenges
A patent can be shortened to zero through a successful challenge — either in inter partes review (IPR) or post-grant review (PGR) at the PTAB, or in district court litigation. Invalidation does not literally change the nominal expiry date, but it ends the monopoly just as surely. Given the high invalidity rate in contested IPR proceedings, the "effective" term of many patents is considerably less than their paper term.
How Does the US Compare Globally?
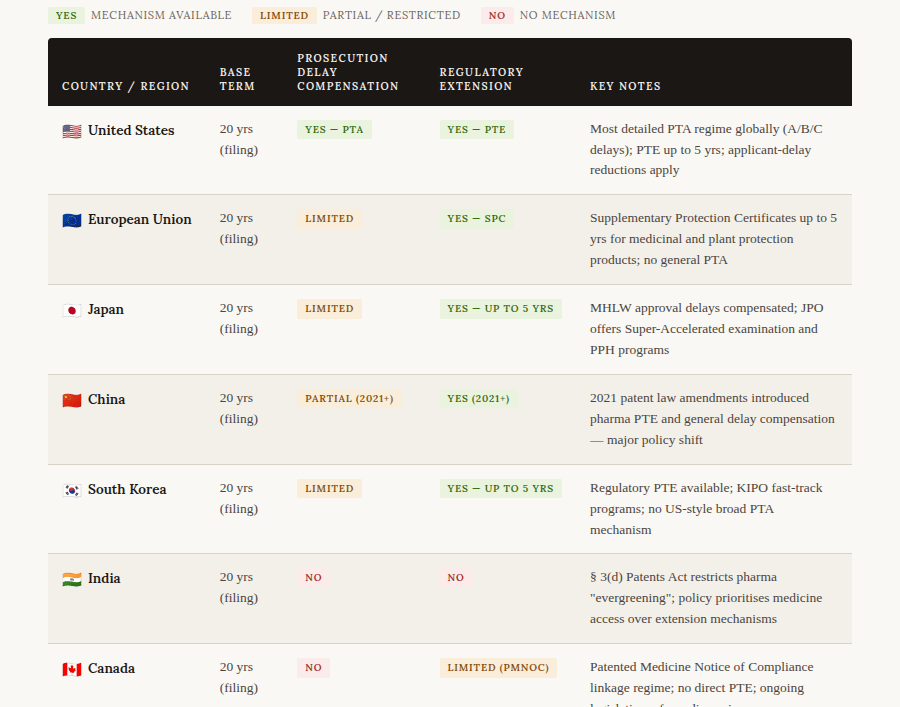
The 20-year-from-filing standard is now the global baseline, mandated by the Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS), which virtually every country in the world has signed. Where countries diverge sharply is in whether and how they compensate applicants for prosecution or regulatory delay.
The most notable recent development in global patent term law is China's 2021 reform. Historically, China offered no mechanism to compensate for either long examination delays at the CNIPA or lengthy NMPA drug approval processes. The 2021 amendments changed this fundamentally, introducing both a patent term compensation system for innovative pharmaceutical products and a more general mechanism to offset unreasonable examination delays. This brings China's framework meaningfully closer to the US and European models, reflecting the country's ambition to be seen as an innovator-friendly jurisdiction.
The European SPC regime deserves special mention. While the EU offers no direct equivalent of PTA for ordinary prosecution delay, the Supplementary Protection Certificate grants up to five additional years of exclusivity for medicinal products and plant protection products that required regulatory authorisation. For blockbuster drugs, SPCs have been some of the most commercially valuable IP rights ever granted, and litigation over SPC scope regularly reaches the Court of Justice of the EU.
India's approach is strikingly different from the rest. Section 3(d) of the Indian Patents Act explicitly bars patent protection for new forms of known substances unless they demonstrate significantly enhanced efficacy — a direct legislative attack on "evergreening," the practice of filing minor variation patents to extend commercial exclusivity beyond the life of the original compound patent. India also offers no PTA or PTE. This philosophical divergence reflects a deliberate policy choice to prioritise access to affordable medicines over the incentive model embedded in Western IP frameworks.
Practical Implications
For an inventor or a company, these rules translate into a set of concrete strategic decisions. Filing a continuation is not free — it accelerates the terminal disclaimer risk and may shorten the effective commercial window. Responding to Office Actions slowly burns PTA that could otherwise compensate for a lengthy examination. Choosing whether to file a US national stage application or to enter via a PCT route affects how the three-year B-delay clock starts. For pharmaceutical companies, the interplay of PTA, PTE, and terminal disclaimer law can mean the difference between a multi-billion-dollar product lifecycle and a generic cliff arriving years early.
As MPEP § 2701 reminds us, the terminal date printed on every patent certificate is calculated from a single day — often one that predates the issued patent by years. Understanding the arithmetic of that gap, and the legal tools available to reclaim some of it, is no longer just a concern for specialist patent attorneys. It is a core element of any serious IP strategy.
Key References
35 U.S.C. § 154 — Duration; Patents and Fees
35 U.S.C. § 156 — Extension of Patent Term
MPEP § 2701 — Patent Term
MPEP § 2731 — Basis for Patent Term Adjustment
MPEP § 2733 — Reduction of Patent Term Adjustment
TRIPS Agreement, Article 33 — Duration of Protection
EU Regulation 469/2009 — Supplementary Protection Certificates
China Patent Law (2021 Amendment), Articles 42 & 76
(Disclaimer: This article is for informational purposes only and does not constitute legal advice. Always consult with a qualified patent attorney regarding your specific situation.)